A spokesperson for the company responded to CBS News, however, saying that Infinant Health is planning to continue distributing its “Evivo powder product” for consumers to buy and intends “to work with the FDA toward approval of the use of our MCT oil product in hospital settings.”
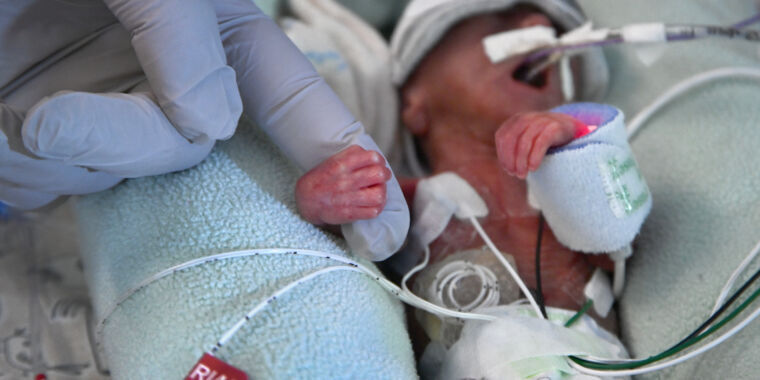
Why the FDA is upset specifically: “Moreover, the agency took aim at the company touting the supplement as “made specifically for use in healthcare settings,” and “[d]esigned specifically for infants in the NICU[.]” NICU is the Neonatal Intensive Care Unit, where the most vulnerable of newborns, many preterm, receive specialized and intensive care.”
And also they claim their study design was bad and inadequate to proof efficacy.
But the article also says 10% of such cases recieve such probiotic products. Would be interesting to see what the outcomes of those 10% are vs the 90%.
C’mon arstechnica, does the FDA need to “blast”? What’s next “WHO slams modern pharma industry”?
It’s well known that infants should never have honey for this reason. Why on earth would you purposefully expose them to this orally?
It seems like the “superfood” advertising concept being applied to formula.
Honey is dangerous because of the risk of botulism. Botulism isn’t mentioned at all in the article, so why do you bring it up?
Specifically because they cannot withstand the spores when ingested, just like here. This isn’t confusing.
Botulism is obviously dangerous. The bacteria here is being introduced to infants with the goal of reducing the risk of a specific condition, although it’s an unlicensed treatment.
I might suggest that off label usage of medication should be avoided with infants.
When you put it like that…
So is it the process by which MCT (medium-chain triglycerides) oil is extracted and added to the formula in this specific product, or is there something about MCT oil which makes it a particularly dangerous growth medium for the bacteria that killed the infant?